Early Cancer Detection
The Process

Abnormal Cell Growth
Target Patient

Benign or Malignant

Tumor Type Classification
Malignant tumors have different shapes, sizes and impacts. Classifying the tumor correctly is important to plan treatment

Timely Diagnosis
Malignant tumors are dangerous and can spread quickly. Timely classification and treatment can save lives
Solution

Model

Training

Deployment
Product
A COMPLETE SOLUTION FOR multiple types of cancer tumor classification using pathological images THAT CONTINUOUSLY LEARNS AND IMPROVES
Digital Image Input
AI Examination
Automated Classification
Universal Access
The solution is accessible globally through the internet
Data Platform
Our data science and engineering teams collaborate with leading research, commercial, and government data platforms to collect large-scale datasets that fuel our machine learning frameworks and AI algorithms. Our robust models and testing systems ensure high accuracy and reliability, allowing us to deploy the most effective features for each use case.
Our processes are fully compliant with global data privacy regulations.
Data Sources Include:
Major research universities
Government and non-government health organizations (CDC, NIH, NHS, etc.)
Public research data repositories (Figshare, Kaggle, etc.)
Commercial health and biometric data sources
Technology Platform
Our platform is built on scalable, robust, and advanced global infrastructure combined with highly skilled data science and software engineering expertise.
Microsoft Azure Services Partner
Operates on Microsoft Azure for big data, machine learning, and AI capabilities
Collaborates directly with Microsoft Azure engineers to ensure high-quality software and services
Our Team
Our data science and software engineering teams include PhDs and master’s degree holders from leading American and international universities.
Methodology
Integrated radiomics
Model Architecture
Integrated radiomics combines X-rays, MRI, CT scans, and other medical imaging data. These inputs are processed through convolutional layers to enable advanced AI-driven analysis, tumor classification, and early cancer detection.
Small receptive field filters are used for linear and nonlinear transformations of input channels
Tailored fully connected layers classify tumors into categories such as meningioma, glioma, and pituitary tumors
The model is refined using Gabor filters and color blob detection
Datasets
Multiple tumor image datasets include thousands of T1-weighted contrast-enhanced images. These datasets are used to train and test the model.
Primary features analyzed include:
Tumor size
Position
Shape
Texture
Datasets are continuously updated and improved.
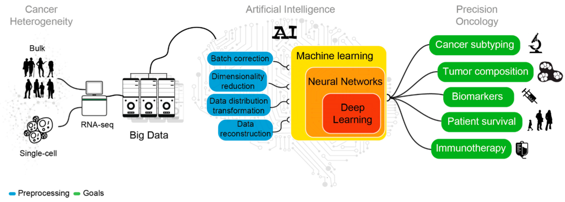
Integrated Multiomics
AI-Driven Biomarker Discovery Using Multiomics Data
Doctari leverages integrated multiomics analysis to identify critical biomarkers for early cancer detection, risk prediction, and personalized diagnosis.
By combining multiple biological data layers—genomics, transcriptomics, metabolomics, and proteomics—our AI provides a deeper understanding of disease development at the molecular level.
Genome Sequencing
One of the indicators of certain types of cancer is the presence and analysis of bone marrow mononuclear cells.
Additional insights can be gained through genome mapping, where both germline and somatic mutations are analyzed to assess cancer risk and disease progression.
Deep learning plays a critical role in clinical oncology by supporting:
Early diagnosis
Prognosis prediction
Identification of cancers of unknown origin
Molecular subtyping of cancers
Precision oncology based on genomic data
This enables more accurate, data-driven decisions in cancer detection, treatment, and long-term patient outcomes.
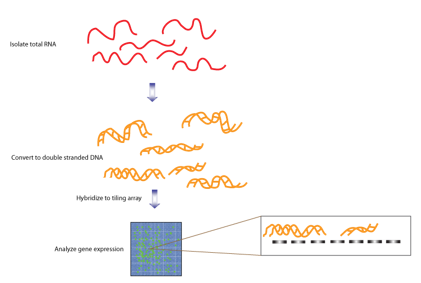
Transcriptome Sequencing
The genome is not the only place where mutations can occur. The transcriptome—particularly RNA—can also influence the development of conditions such as MDS and AML. This area is less explored than genomics, and AI has the potential to drive new discoveries.
Transcriptomics-based machine learning can:
Predict AML status without requiring expert input
Support diagnosis and subclassification
When combined with machine learning as part of an integrated multiomics approach:
Genomics enables risk prediction, differential diagnosis, and subclassification
Transcriptomics supports diagnosis through AI-driven analysis
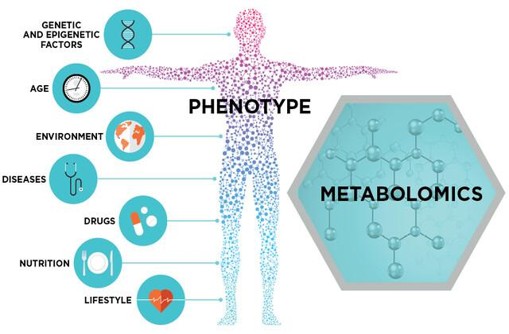
Metabolomics
Metabolomics focuses on small-molecule metabolites in the blood, including amino acids, lipids, and carbohydrates. Over 5,000 metabolites—both identified and unidentified—can be analyzed using AI.
Metabolomics data can be used to:
Discover unknown metabolic pathways and their regulation
Identify preferred metabolic routes within biological systems
Predict the inhibitory effects of substances on metabolic networks
Machine learning, combined with logic-based modeling (including abduction and induction methods), can help predict potential inhibitory side effects of drugs within metabolic systems.
Proteomics
Proteomics studies proteins such as cytokines and chemokines, which are secreted into the bloodstream by the immune system and play a vital role in fighting disease. Certain pro-inflammatory protein patterns have been associated with MDS symptoms.
Proteomic data is used to:
Train machine learning predictive models
Support both regression-based and neural network forecasting
Enable prognosis based on protein signatures of MDS or AML patients
Future validation of these models in real-world settings could enable proactive monitoring of patient health.
Digital biomarker detection
Digital pathological images combined with multiomics data are used for analysis, achieving up to 93% accuracy in detecting, classifying, and labeling multiple types of cancer.
Communication between client software and cloud-based systems is secure
Tumor classification reports can be viewed on any device or exported as PDFs
Physicians and researchers can make more informed decisions based on detailed tumor insights
Continuous feedback helps improve the system over time
